Pour point
The pour point of a liquid is the lowest temperature at which a sample of the fluid shows flow characteristics under defined conditions. Especially at crude oils and oil products it must be considered that these fluids may show a thermal history.
The knowledge of the pour point of a fluid is especially at fluid transportation of big importance. A solidifying fluid can plug pipelines and so pumps can be damaged by overstressing or the feed rate is strongly decreasing.
In petroleum industry many standards are in use, due to the history of petroleum industry the standards of ASTM International1 (originally: American Society for Testing and Materials) are dominant.
For pour point measurement the important standards are:
- ASTM D97 (for petroleum products) and ASTM D5853 (for crude oils), both using the tilt method
- ASTM D5950 (automatic tilt method)
- ASTM D5985 (rotational method)
- ASTM D7346 (no flow point, air pressure method)
These methods differ mainly in their accuracy, the needed expenditure of time and sample amount.
Measurement method according to ASTM D5985
According to measurement method ASTM D5985 (Standard Test Method for Pour Point of Petroleum Products (Rotational Method)) the sample cup is filled with the test specimen (grafic: 3), then the cup is set to a slow rotation of about 0.1 rounds per minute (4). The coaxial, tiltable bedded temperature sensor (2) is dipping in the sample fluid. When the pour point is reached the viscosity of the sample increases and by this the temperature sensor is moved out of its position and triggers the light barrier (1).
1. Light barrier | 2. Temperature sensor | 3. Test specimen | 4. Motor (~0,1 rpm) |
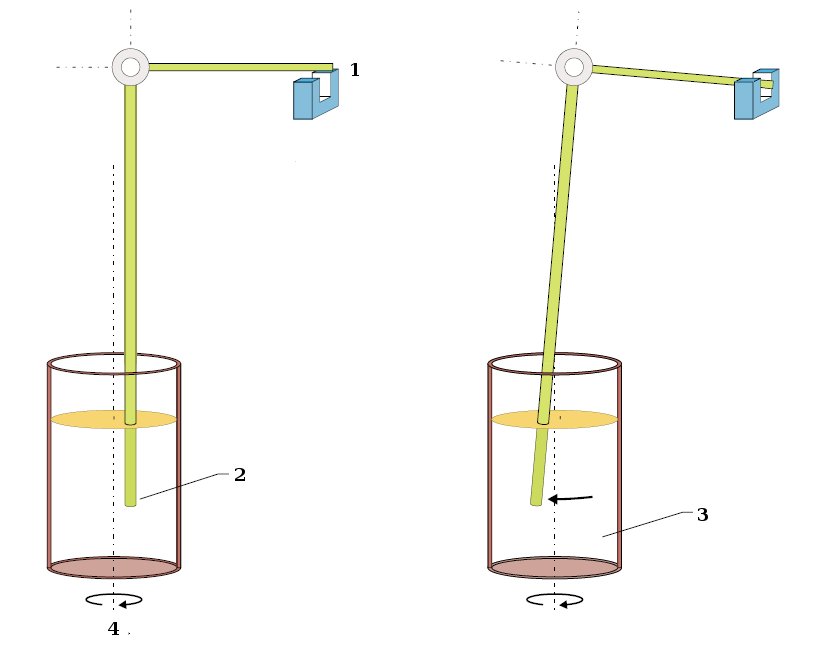
Measurement method according to ASTM D97 / D5853
According to ASTM D97 (for petroleum products) respectively ASTM D5853 (for crude oils) the pour point is obtained as follows: the test specimen is heated until it is clearly capable of flow. In a test jar the sample is cooled down slowly in steps of 3 K. Every 3 K the flow behaviour is controlled by tilting the test jar. If the sample shows flow behaviour it is cooled down another 3 K. If the sample shows no flow behaviour anymore the temperature (in °C) is rounded up to the next higher by three divisable (without remainder) integer, this is the pour point according ASTM D97 / D5853.
Comparison of different measuring methods
In the following the different measuring methods for pour point determination are compared. The reference is the ASTM D5985 (Rotational Method) which is used in the device Pour Point Tester PPT45150 by PSL Systemtechnik GmbH.
- With the rotational method the pour point can be determined with an accuracy of 0.1 K. The temperature of the sample is measured directly inside the sample, hence the thermal behaviour of the test specimen can be observed during cooling down. This gives with one measurement informations about crystallisation effects. During crystallisation heat energy is released, this slows down the cooling. By using graphical or computer-assisted analysis the change of the cooling curve can give evidence about the cloud point of the sample or at crudes and heavy oils about the WAT (Wax Appearance Temperature). The sample is not mechanically disturbed by this measuring method, this increases the reproducibility of measuring values.
- At the manual tilt methods (ASTM D97 and D5853) as well as at the automatic tilt method (ASTM D5950) the samples are tilted every 1 or 3 K and by this strongly mechanically disturbed. This can influence the crystallisation process and reduce the reproducibility. Also is the accuracy limited to 1 or 3 K, depending on the used method. The cooling rate is depending on the ASTM specifications (air cap between sample vessel and cooling mantle) relatively slow, the expenditure of time for a measurement increases. Moreover no information about the thermal behaviour of the sample can be obtained depending on the discontinuous data acquisition.
- At the test method ASTM D6749 (Automatic Air Pressure Method) the surface of the sample is tested with pressurised air every 1 or 3 K (depending on measuring device). This disturbes the crystallisation process unless this disturbance is not as effecting as the tilt methods. Also the accuracy is depending on the test limits at 1 or 3 K. Here, too, no information about the thermal behaviour of the sample can be obtained.
- According to ASTM D7346 (Pressure Differential Method) the pressure change of a closed area over the test specimen is measured. Crystallisation of the sample induce a pressure change in the test chamber, the no flow point is obtained. This point is about 0.1 K below the pour point. The accuracy of this method is about 0.1 K and so it is comparable to the rotational method. But the real flow characteristic can sometimes not be obtained because this method is reliant to a density change during crystallisation. If such a density change is missing or if several changes in alloys are interfering this method can generate strongly differing values for pour points compared to other methods. The advantages of this measuring method are the very small amount of sample of less than a millilitre needed to perform a test and the short measuring time.
Freezing point and viscosity
The freezing point marks the temperature TG at which a substance turns from liquid to solid state at a certain pressure. This point is a constant for a pure substance. By impurities the freezing point is always moved to lower temperatures (freezing point depression2, cryoscopic constant). The depression depends only on the amount, not the nature of soluted substance (colligative property3). The pressure dependence is small compared to the pressure dependence of the boiling point but it can be well observed at high pressures (phase diagrams of substances without anomaly).
Freezing is a form of crystallisation. The molecules change from a unsorted, chaotic liquid phase to a more or less sorted crystal structure. This phase transformation is bounded with releasing of energy, the crystallisation enthalphy which is emitted as heat. This heat energy is contrary to the external cooling. In an ideal process this leads to a constant temperature during the whole crystallisation. The temperature of the substance decreases not until this process is finished.
In multi phasic alloys only in rare cases (eutectic mixtures) all components crystallise at the same temperature. Normally only one component crystallises, the formed crystals exist as colloids in a solution. Due to the Tyndall effect 4 (light scattering at the phase interface of colloids) the solution becomes cloudy (cloud point). When growing the crystals bond to a three-dimensional structure and due to this the flowability of the solution decreases, the viscosity increases.
In solutions of complex composition like crude oil or petroleum products often dozens of substances are combined. This gives multiple interfering solution and crystallisation equillibria. These alloys normally produce mixed crystal systems.
By forming three-dimensional structures the viscosity of the compound increases more than only by simple cooling. The flowability is increasingly reduced until it is finally evanescent, the fluid freezes.
Viscosity
The viscosity is the resitance of a fluid against shear stress. For fluids it is differed between the dynamic viscosity η and the kinematic viscosity ν which is the dynamic viscosity divided by the density ρ of the fluid.
Footnotes:
(Links will open in new window)
- Main page ASTM International
- Freezing point depression (Wikipedia)
- Colligative Properties (Wikipedia)
- Tyndall effect (Wikipedia)


